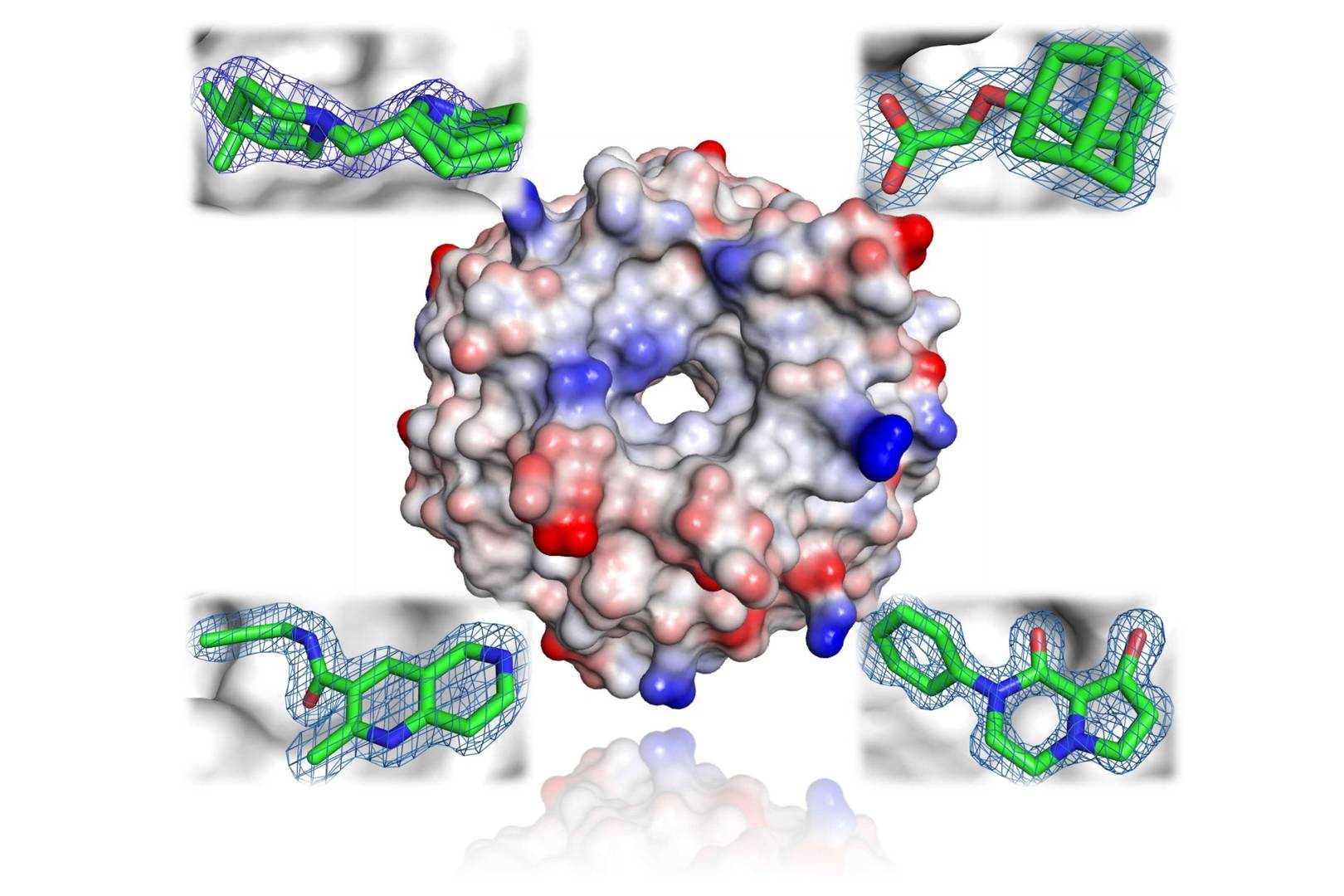
Nuclear magnetic resonance (NMR) spectroscopy is essential in structural biology for revealing the 3D structures of proteins, nucleic acids and their complexes. It provides detailed insights into molecular dynamics, interactions and conformational changes. NMR aids in validating target engagement, identifying and characterising chemical compounds confirming initial hits.

At Nuvisan, we leverage our extensive expertise and advanced NMR capabilities to build confidence and guide your project. We offer NMR-based fragment screening, from identifying initial hits to conducting confirmation and analogue studies, ensuring comprehensive support. Additionally, we provide early biophysical verification of ligand binding to cryo-EM protein constructs under conditions similar to cryo-EM environments.


Our structural biology team delivers high quality 3D information of your compounds of interest to guide the design of improved chemical matter.
learn more
We support you with in-depth analysis of compound-target interactions with our biophysics toolbox.
learn more